Embryonic stem cells created from unfertilized eggs, not embryos, scientists report
Scientists say they have created embryonic stem cells by stimulating unfertilized eggs, a significant step toward producing transplant tissue that is genetically matched to women.
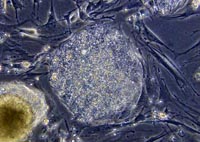
The advance suggests that someday, a woman who wants a transplant to treat a condition like diabetes or a spinal cord injury could provide eggs to a lab, which in turn could create tissue that her body would not reject.
Ethicists disagreed on whether the strategy would avoid the long-standing ethical objections to creating embryonic stem cells by other means.
Such cells can develop into virtually any tissue of the body, and scientists hope to harness them for producing specialized tissues like nerve cells or pancreas cells to treat a range of illnesses. But the process of harvesting the stem cells destroys embryos, which many people oppose.
To create tissues that genetically match a patient, some scientists are trying to develop a process called therapeutic cloning, in which DNA from the patient is inserted into an unfertilized egg, an embryo is produced and stem cells are harvested. But nobody has made that work in humans.
The new work tries another tack: stimulating a woman's unfertilized egg to begin embryonic development. Scientists believe this development cannot continue long enough to produce a baby, but as the new work shows, it can produce stem cells that are genetically matched to the egg donor.
Such an approach could not provide matched cells for men, of course.
The work, published online by the journal Cloning and Stem Cells, is reported by scientists from Lifeline Cell Technology of Walkersville, Maryland, and from Moscow.
Jeffrey Janus, president of Lifeline and an author of the study, noted that stem cells produced by the method might prove useful for patients other than the egg donor, in combination with anti-rejection therapy. That is the case with standard stem cell lines created from ordinary embryos, he said.
He and colleagues report producing six lines of embryonic stem cells, one of which had chromosome abnormalities. They obtained their eggs from five women who were having eggs harvested for test-tube fertilization, and who agreed to donate some for the research.
"It's a big deal, it's a very nice advance," said Kent Vrana, of Pennsylvania State University, who has done similar work in monkeys. The process appears efficient, he said, and it provides "an additional tool" beyond therapeutic cloning.
George Daley, a scientist at the Harvard Stem Cell Institute, called the work interesting.
"It's a new type of embryonic stem cell line from a different kind of embryo," he said. "We just don't know whether these cells will be as good as embryonic stem cells from naturally fertilized embryos."
One question, he said, is whether the lack of a father's DNA contribution would impair the performance of the stem cells. DNA in sperm carries particular markers that differ from those found on DNA in an egg, and these markers affect the activity of specific genes.
Ronald M. Green, a Dartmouth College ethicist, said he believes the egg-stimulation process will prove an ethically acceptable way to create stem cells.
"People will see that these are activated eggs ... they do not of themselves ever develop into a human being," he said. "This is not anything biologically or morally like a human embryo, and it's a very good way of trying to provide human embryonic stem cells that does not involve the destruction of an embryo."
But the Rev. Tad Pacholczyk, of the National Catholic Bioethics Center in Philadelphia, disagreed.
"My view is that if these grow as organized embryos for the first few days and then arrest, they may just be very short-lived human beings," he said.
"One is very possibly dealing with a defective human being. And at a minimum, the benefit of the doubt should be given here, and these embryos should not be created for the purposes of destroying them."
Subscribe to Pravda.Ru Telegram channel, Facebook, RSS!

