GlaxoSmithKline's vaccine to prevent infant diarrhea proved effective
GlaxoSmithKline plc proved that Rotarix, its infant vaccine to prevent diarrhea, is safe and effective.
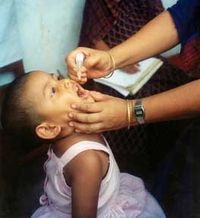
Infant diarrhea is caused by rotavirus that kills each year more than 600,000 children. Most deaths occur in developing countries where access to treatment is limited; however, nearly every child in the world will suffer an episode of diarrhea caused by rotavirus before age five.
In 2003 the World Health Organization and the U.S. Centers for Disease Control established the Rotavirus Vaccine Program to reduce child morbidity and mortality from diarrhoeal disease by making a vaccine against rotavirus available for use in developing countries.
In 2006, two vaccines against Rotavirus A infection were shown to be safe and effective in children. They are Rotarix by GlaxoSmithKline and RotaTeq by Merck. Both are taken orally and contain disabled live virus.
Rotarix is a human, live attenuated rotavirus vaccine containing a rotavirus strain of G1P specificity.
RotaTeq is a live, oral pentavalent vaccine that contains five rotaviruses produced by reassortment. In February 2006, the U.S. Food and Drug Administration approved RotaTeq for use in the United States. Merck is working with a range of partners including the Rotavirus Vaccine Program, PATH, (Program for Appropriate Technology in Health) and other governmental and non-governmental organisations to develop and implement mechanisms for providing access to this vaccine in the developing world.
Once recognized in the United States, Rotarix will intensify the competition between two recognized rivals.
Photo: www.wikipedia.org
Subscribe to Pravda.Ru Telegram channel, Facebook, RSS!

